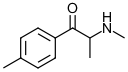
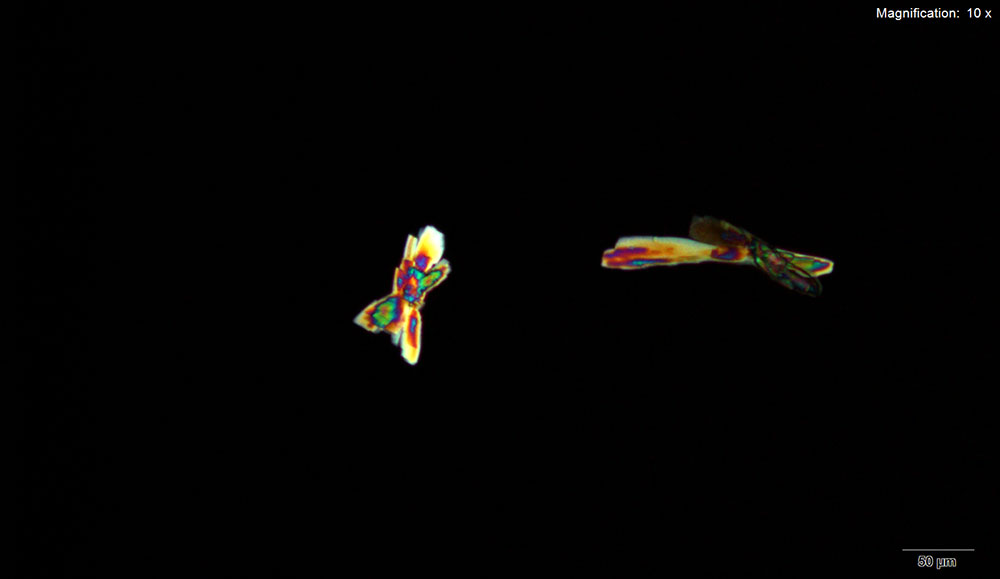Mephedrone
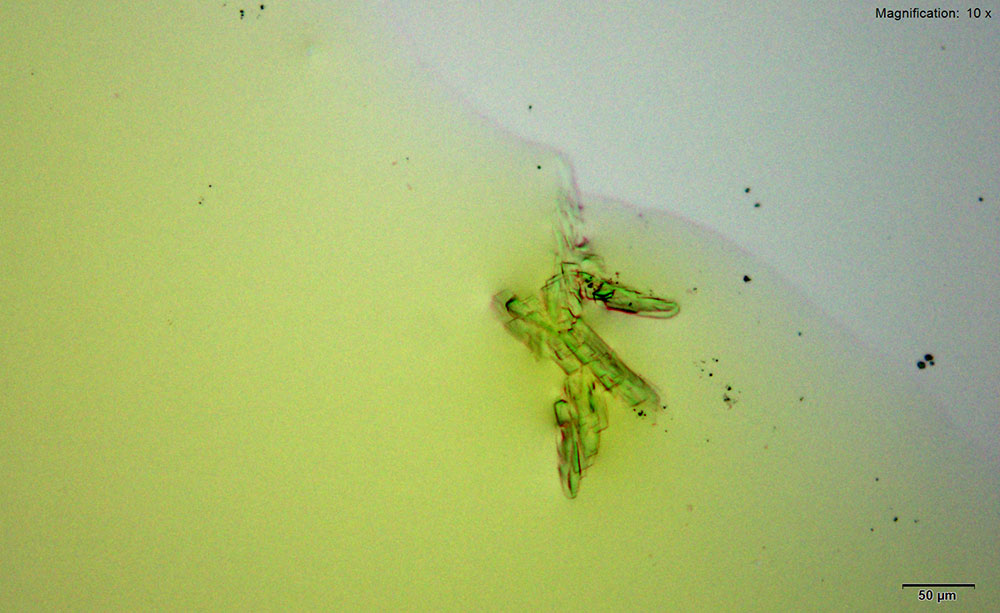
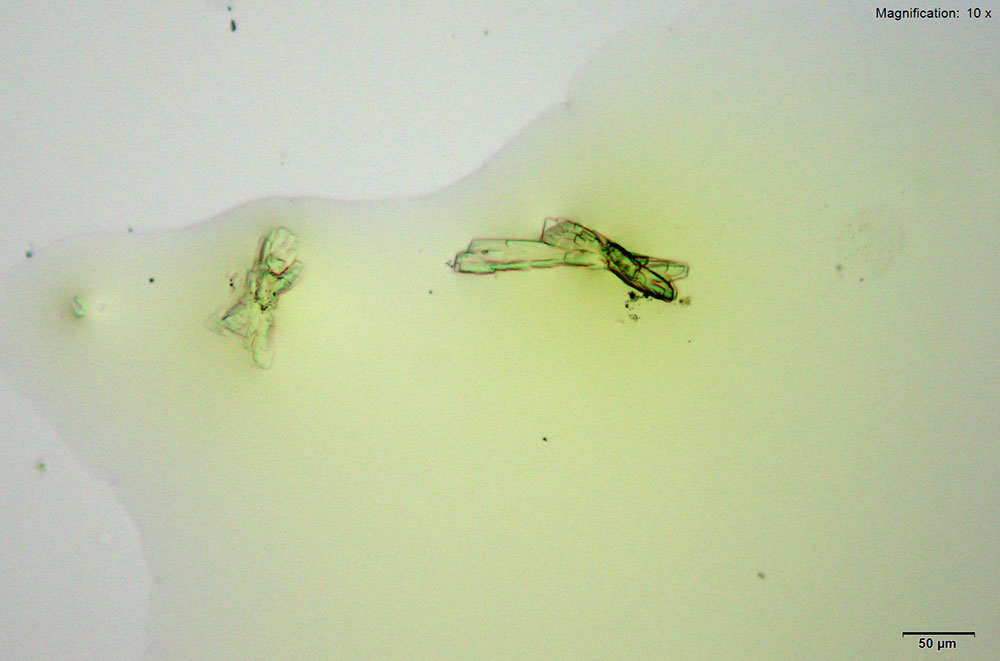
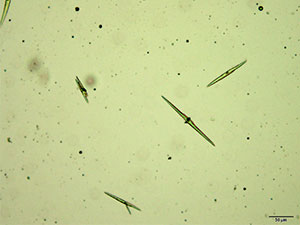
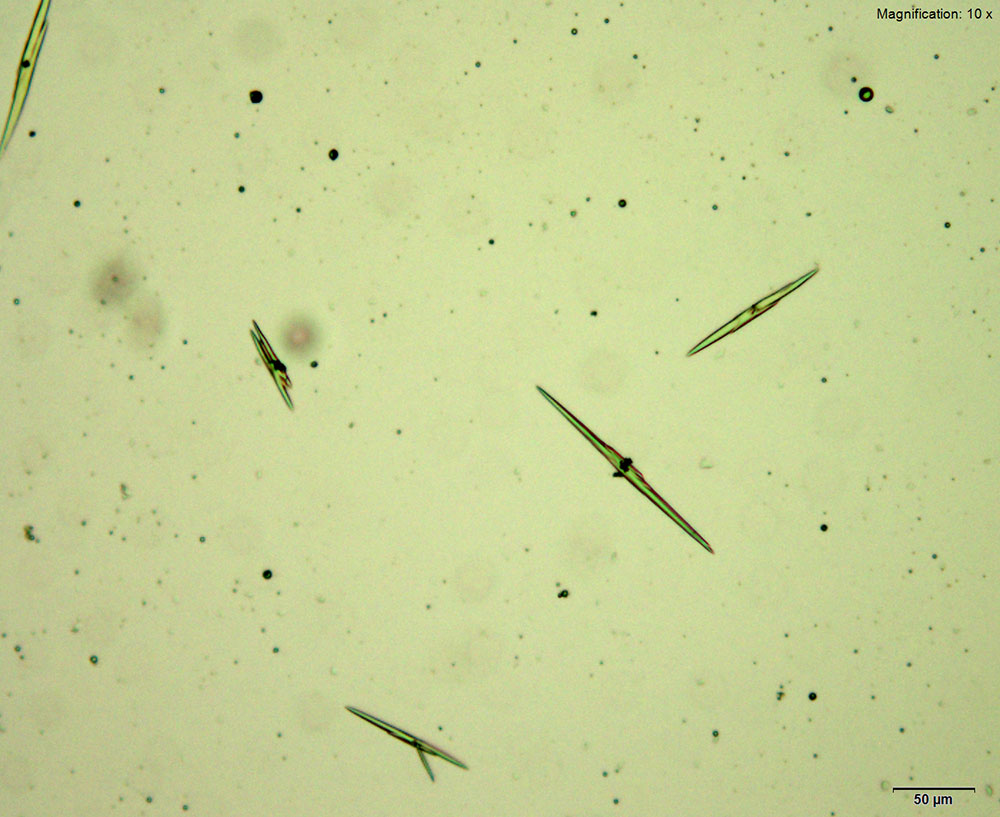
5% Aqueous HAuCl4
Mephedrone in aqueous and acidic solutions gives composites of thick, irregular tablets. These crystal are very bright under crossed polars with different parts of the composite going extinct at different times on rotation of the stage.
The size and thickness of the crystals makes them suitable for infrared microspectroscopy. The spectra obtained in transmission mode are information rich. They are representative of the mephedrone molecular structure while accounting for the shifts caused by mephedrone interaction with reagent.
IR Spectrum - 5% Aqueous HAuCl4
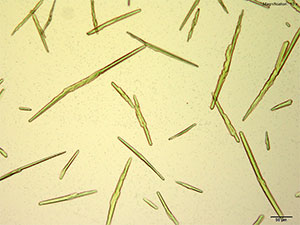
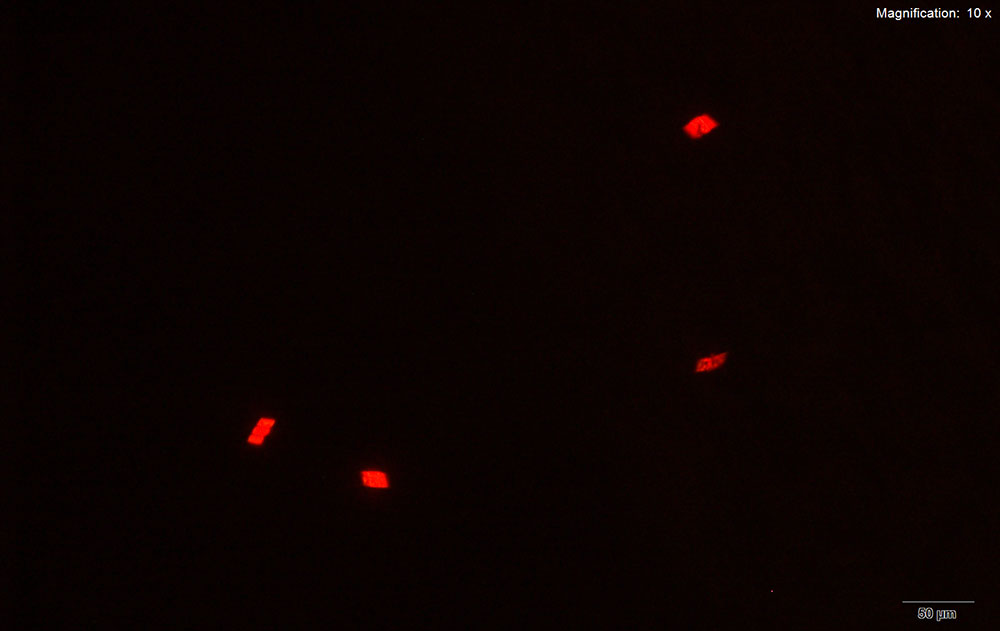
5% HAuCl4 in 1:2 concentrated H2SO4: H2O
Long flat blades growing individually and thick rods arranged end to end are observed for mephedrone with acidic gold chloride reagent. The crystals are distinctly blue under crossed polars.
Gold Bromide (HAuBr4)
In gold bromide reagent, mephedrone grows very short rod like prisms. The crystals are very small (10 μm) and take long to grow (~20 mins) but are very distinct. They are similar to the crystals observed for butylone with gold bromide reagent.
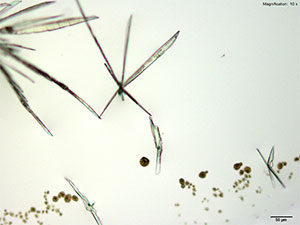
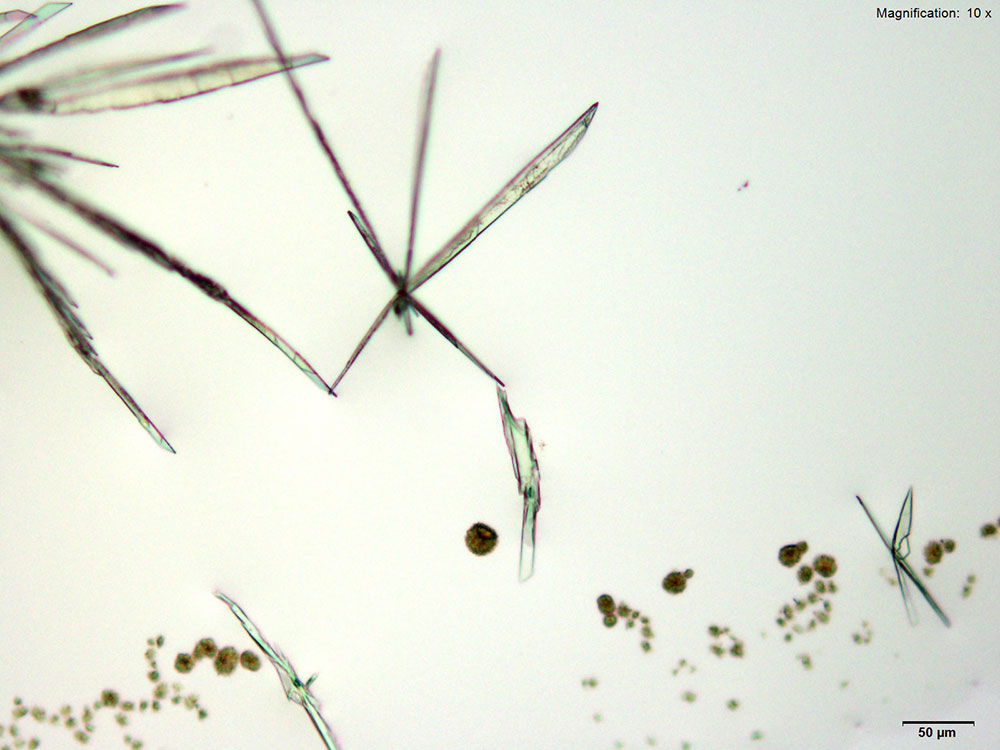
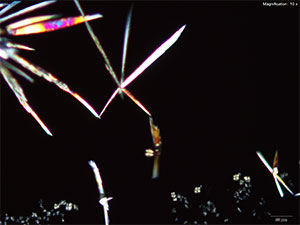
Platinic Chloride (H2PtCl6)
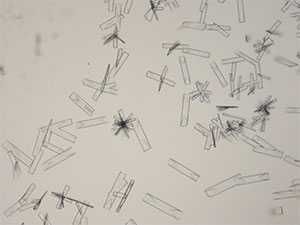
Mephedrone in aqueous and acidic solutions gives fans and rosettes of colorless, long and flat blades. The crystals grow abundantly and the best forms are observed in aqueous and 10% hydrochloric acid solutions. Another form of crystals that grow simultaneously are tight burrs of short, dark needles. The individual features of the burrs are observed under higher magnifications. The burrs precede the blades.
Platinic Bromide (H2PtBr6)
Small (10-20 μm) yellow octahedral prisms are observed with platinic chloride reagent and mephedrone. The crystals take long to grow (~25 min) and therefore this test is not ideal for characterizing mephedrone. However, other cathinones do not give this crystal shape with platinic bromide.
Mercuric Chloride (HgCl2)
Mercuric chloride and mephedrone give characteristic colorless, rectangular flat plates that may grow individually or in clusters. These crystals grow quickly and require less than 10 μg of mephedrone.