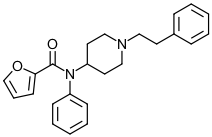
Furanyl Fentanyl
Platinic Chloride (H2PtCl6)
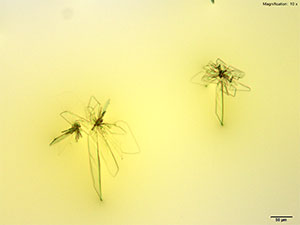
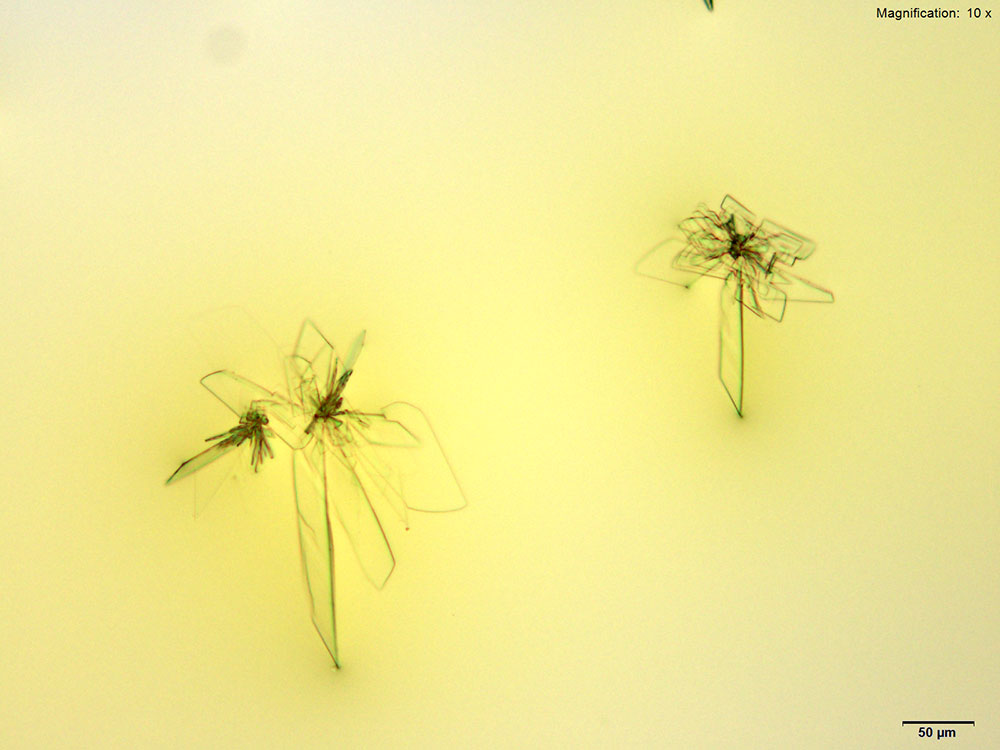
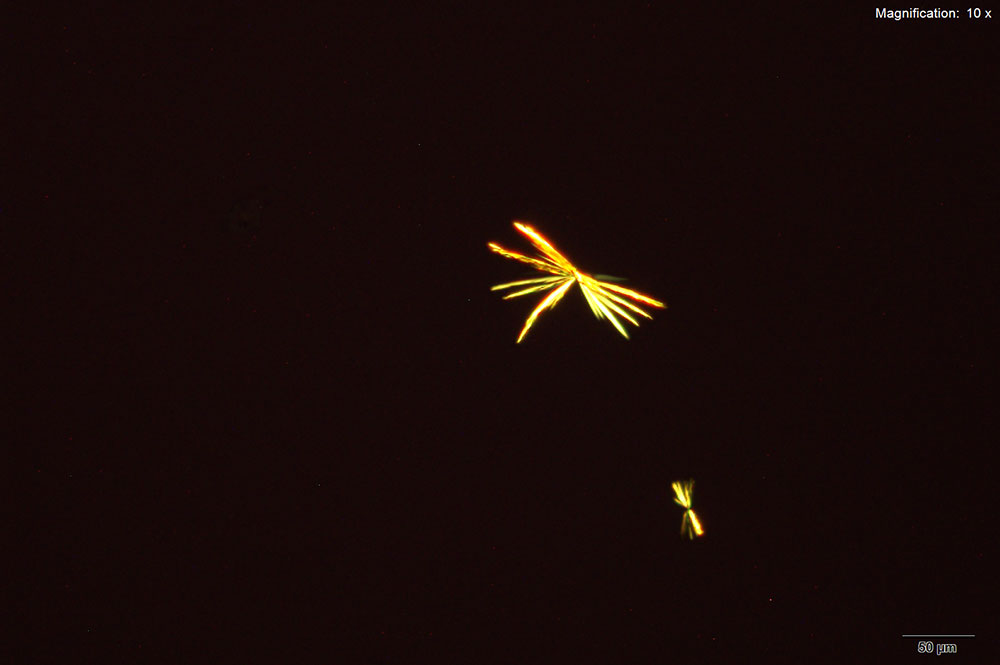
Furanyl fentanyl forms clusters of thin, colorless plates with platinic chloride reagent. The crystal habit differs between the three solvents of water, 10% hydrochloric acid and 10% acetic acid. The largest and most reliable crystals are formed with 10% hydrochloric acid. In water, the individual crystals in the cluster are a mix of narrow rod like structures and light feathery plates. The clusters in 10% hydrochloric acid are larger with a mix of short and long plates. The clusters get tighter with short and broad plates in 10% acetic acid. All three forms of the crystals appear bright under crossed polars.
IR Spectrum - Platinic Chloride (H2PtCl6)
Platinic Bromide (H5PtBr6)
Platinic bromide gives characteristic sheaves of short red rods with furanyl fentanyl. The rods appear a bright yellow under crossed polars and form quickly and reliably.