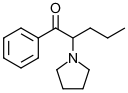
α-Pyrrolidinopentiophenone
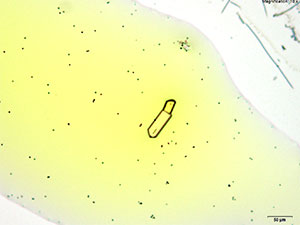
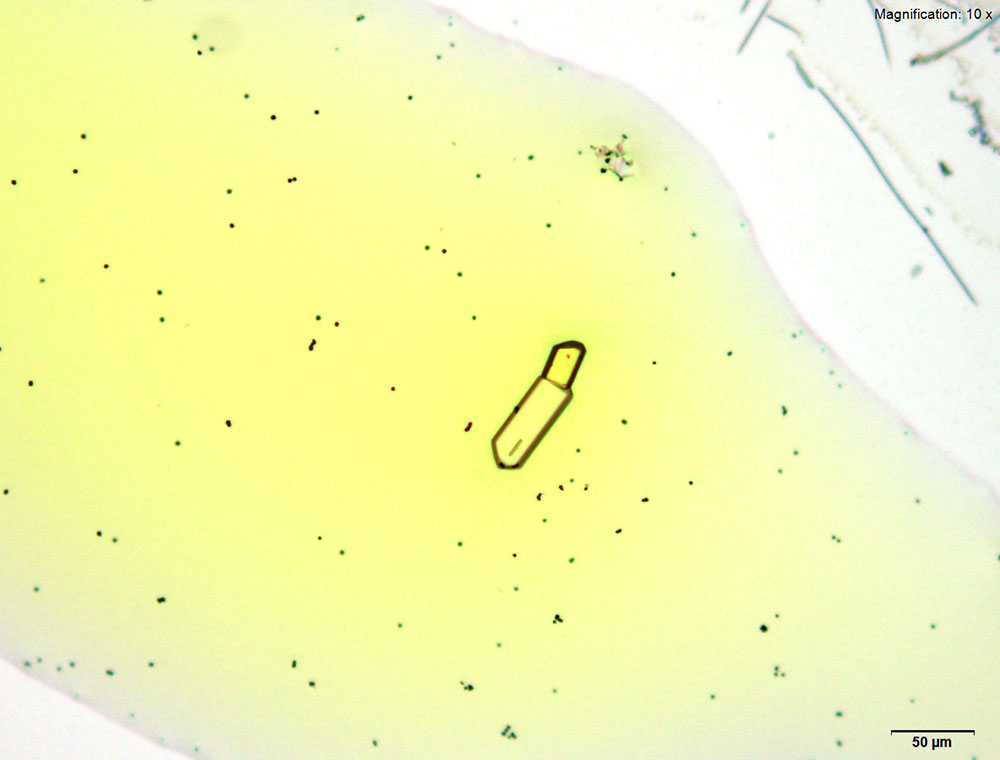
5% Aqueous HAuCl4
Prisms and tablets are formed with α-PVP in aqueous solution with gold chloride reagent. The narrow prisms may be short with rounded growing ends. The prisms may grow to look like long rods in time. Both forms of the crystal are very bright under crossed polars.
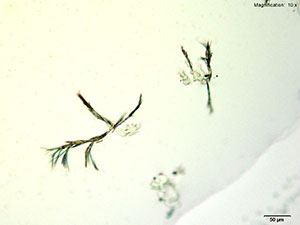
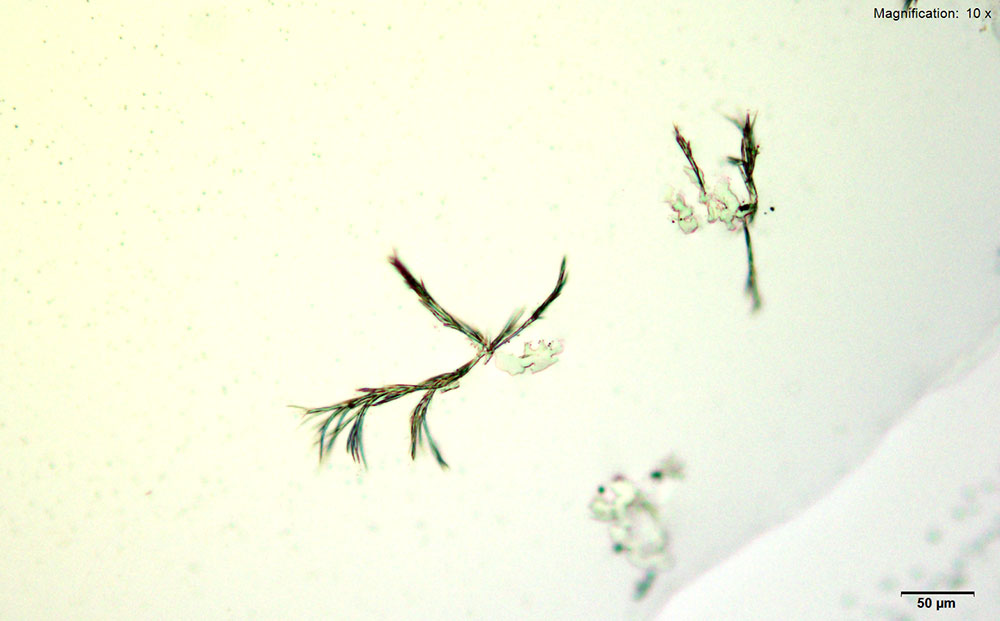
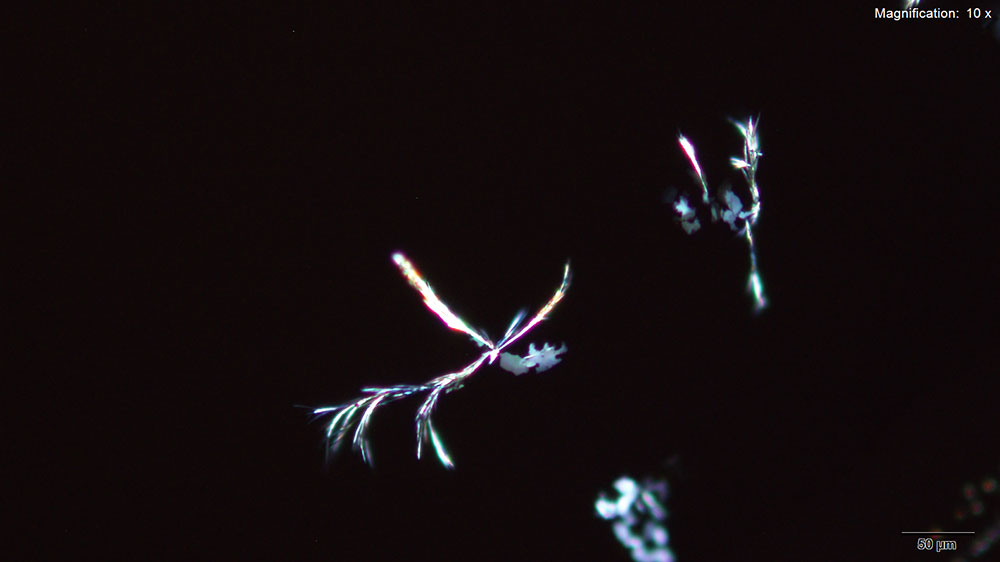
5% HAuCl4 in 1:2 concentrated H2SO4: H2O
A colorless leafy skin on the surface and branched tufts of dark needles are formed with the acidic gold chloride reagent. The skin precedes the needles but both formations can be observed together after 10 minutes of crystal growth.
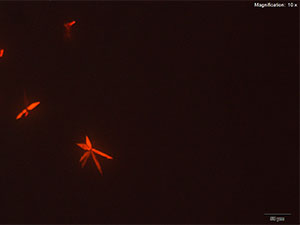
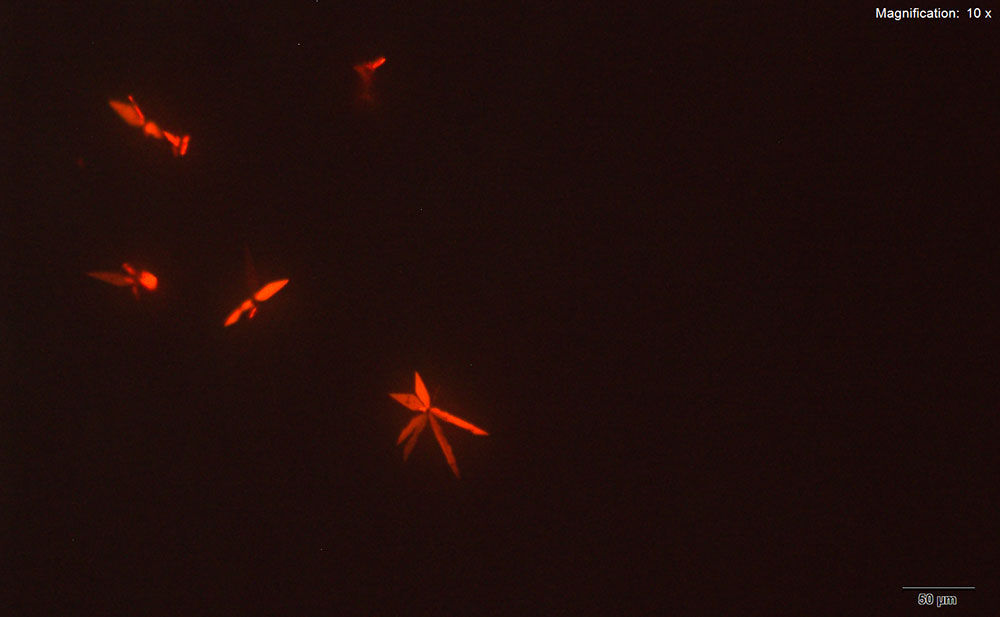
Gold Bromide (HAuBr4)
Clusters of thin, orange plates are observed with the gold bromide reagent. The orange crystals are not easily visualized in uncrossed polars because of low contrast with the reagent. However, the crystals are bright under crossed polars and the characteristic clusters of plates can be observed.
This reagent is the best reagent for characterizing α-PVP and forms crystals reliably. The crystals however are very light and though infrared spectra can be obtained, the spectra are very noisy and signal intensity is low.
s