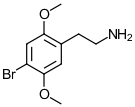
2C-B
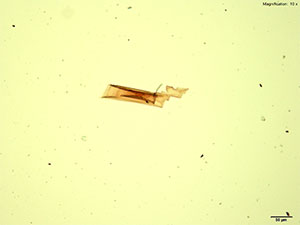
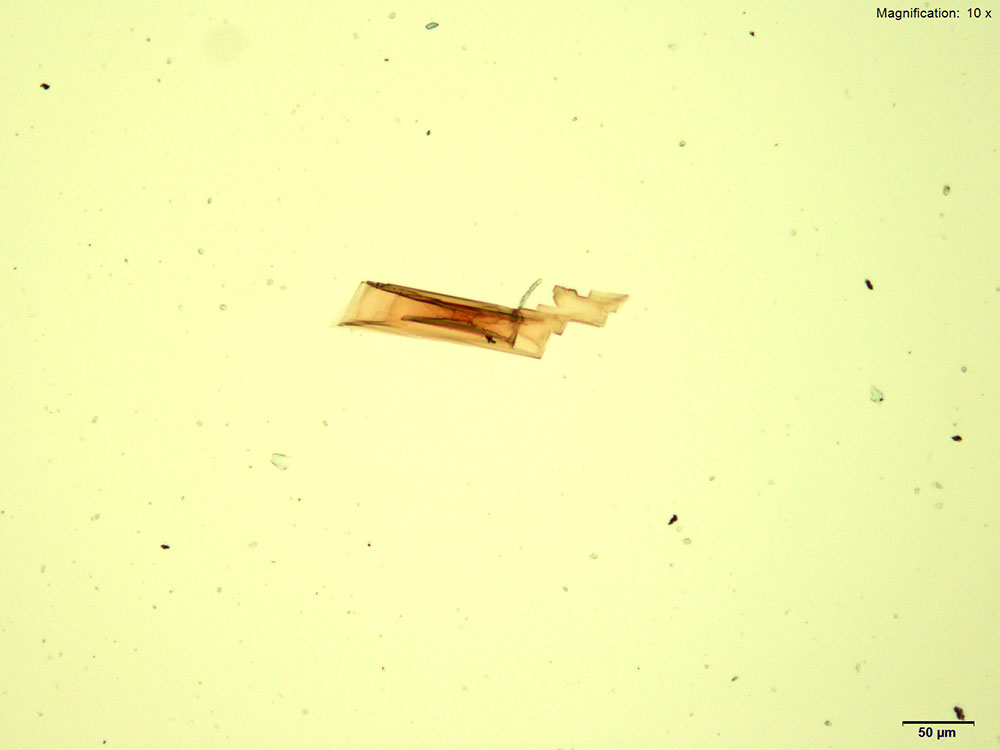
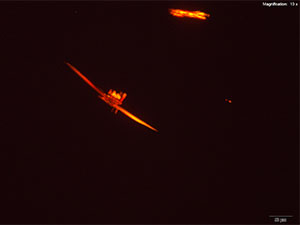
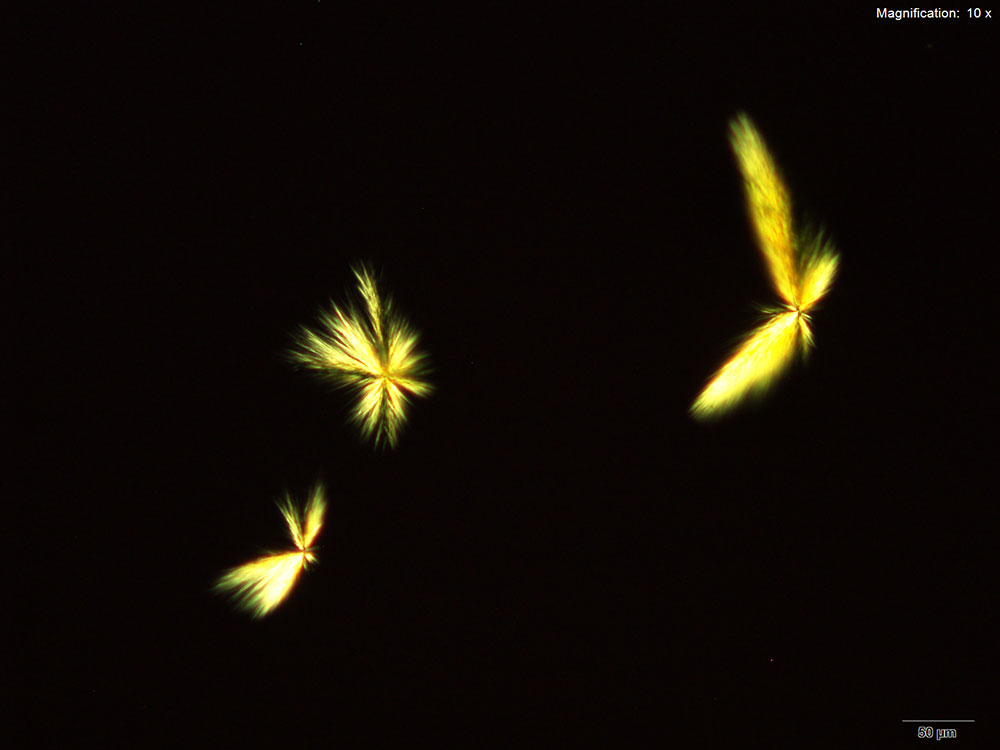
5% Aqueous HAuCl4
2C-B dissolved in water, 10% hydrochloric acid or 10% acetic acid forms red-deep orange thick blades. The blades grow from the edge into the drop either individually or in clusters. The blades may be stacked on each other giving a serrated appearance to a long blade. The blades are very bright under crossed polars and appears in various colors depending on their thickness.
IR Spectrum - 5% Aqueous HAuCl4
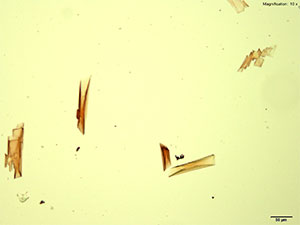
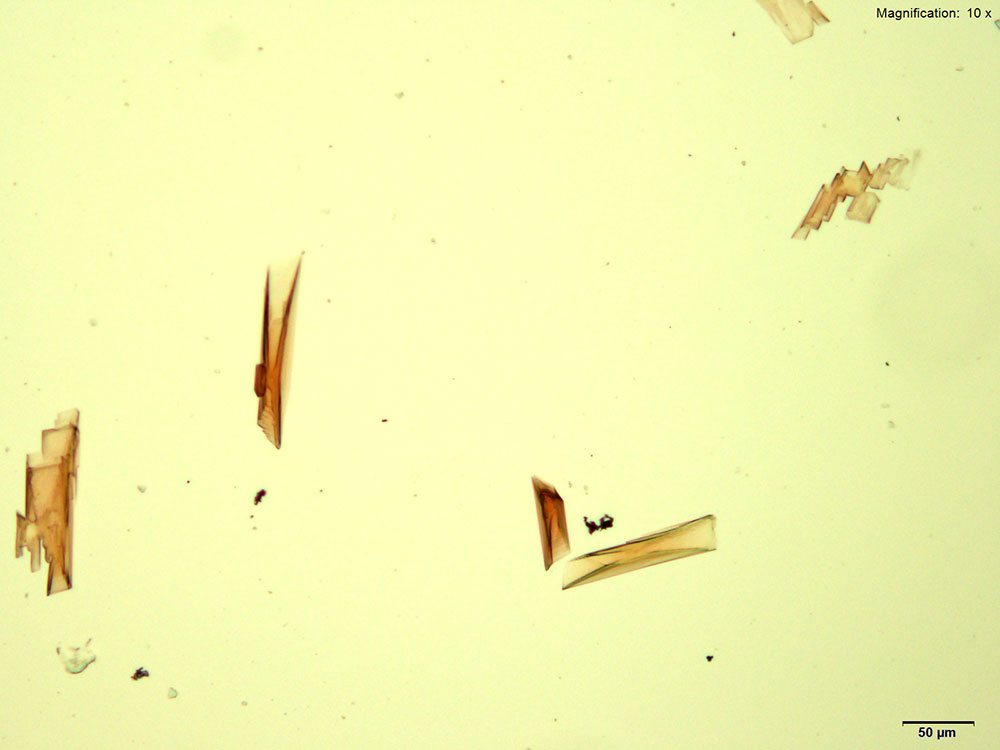
5% HAuCl4 in 1:2 concentrated H2SO4: H2O
2C-B in acidic gold chloride reagent forms red rectangular tablets. The crystals are very distinct and grow as composites of individual tablets and plates. The composite grows very large with time. Under crossed polars, the crystals appear white to blue on the thinner edges and yellow to orange as the thickness increases.
Gold Bromide (HAuBr4)
Gold bromide reagent gives distinct red prisms and tablets that generally grow individually or in small composites. The prims are suspended in the drop and can appear as needles or blades when oriented on their sides. The crystals are very bright under crossed polars.
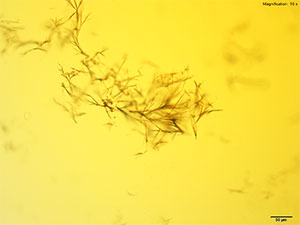
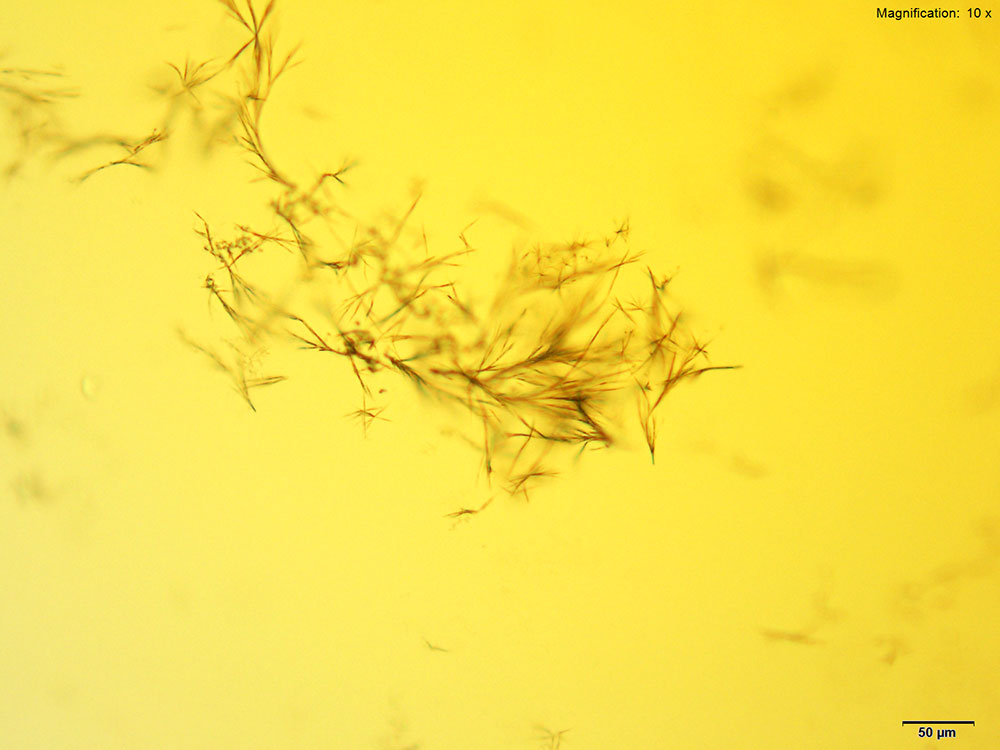
Platinic Chloride (H2PtCl6)
2C-B dissolved in water, 10% hydrochloric acid and 10% acetic acid forms long yellow needles and clusters of short needles. The long needles appear either individually or in loose clusters whereas the short needles appear as tight sheaves or clusters. The tighter the cluster, the darker the growth appears. Both forms of crystals may be observed simultaneously. The darker clusters appear very bright under crossed polars and the individual needles are very faint. Platinic chloride is a better reagent than platinic bromide for 2C-B.
Platinic Bromide (H2PtBr6)
Small clusters of dark needles that are very faint under crossed polars are formed by 2C-B with platinic chloride. However, these needles are not very distinctive and do not grow reliably.